Hexane and isooctane are both nonpolar molecules isooctane actually has a very small dipole moment but it is so small that it can be ignored. But it is not enough to dissolve due to non-polar hydrocarbon group.
File Acetone Dipole Dipole Svg Wikipedia
All alkyl halides are insoluble in water.
. Dispersion Dipole-Dipole Hydrogen-bonding Ion-Dipole. Ion-dipole forces are generated between polar water molecules and a sodium ion. The oxygen atom in the water molecule has a slight negative charge and is attracted to the positive sodium ion.
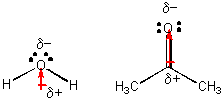
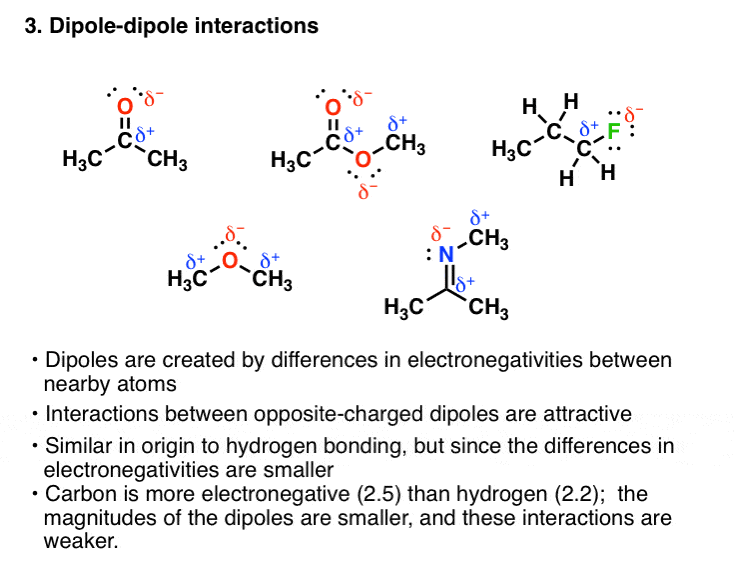
These are typically listed in order of strength. Benzene is not soluble in water because benzene cannot make strong intermolecular forces such as hydrogen bonds or dipole dipole interactions. Dipole-Dipole A dipole force occurs when the positive end of a polar molecule is attracted to the negative end of another polar molecule CH 3 COCH 3 propanone.
D dispersion forces dipole-dipole interactions and hydrogen bonding C dispersion forces and dipole-dipole interactions Ammonia and hydrogen fluoride both have unusually high boiling points due. Alkyl halides can form dipolar - dipolar interactions. Intermolecular forces IMFs are attractive interactions between molecules.
They lead to differences and sometimes trends in various physical properties. Hence the predominant intermolecular forces in both liquids are London dispersion forces. Van der Waals forces Van der Waals forces account for the attraction of the shifting electron-rich portion of one molecule to the shifting electron-poor portion of another molecule.

Intermolecular Forces And Solutions
Which Of The Following Can Hydrogen Bond With Itself Socratic
The Four Intermolecular Forces And How They Affect Boiling Points

0 Comments